New publication about the immune receptor CD300e signaling pathway
Pubblicato il: 04.11.2020 14:48
NEW PUBLICATION: The immune receptor CD300e negatively regulates T cell activation by impairing the STAT1-dependent antigen presentation
Helicobacter pylori is a bacterium that colonize the gastric tract of about 50% of human population. The lifelong permanence of H. pylori in the human stomach suggests that the host immune response fails to clear the infection.
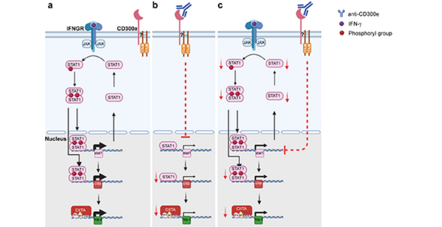
Recently, in de Bernard’s lab was found that H. pylori infection up-regulates the immune receptor CD300e expression in macrophages and that the cross-linking of CD300e affects the ability of macrophages to express the antigen presenting molecule MHC-II, compromising the potential of macrophages to kill the bacterium. However, the mechanism leading to the decline of MHC-II by CD300e activation was not clear.
In the paper published by Coletta S. et al. in Scientific Reports, the authors characterized the pathway elicited by CD300e and demonstrated that the immune receptor compromises the ability of monocytes to activate a specific T cell response, affecting the MHC-II synthesis.
Notably, they found that this effect, that is associated with the transcription impairment of STAT1, overcomes the capacity of IFN-γ to promote the expression of the antigen-presenting molecules.
Taken together, these findings suggest that CD300e may be a novel immune check point that contributes to the regulation of the physiological as well as pathological expansions of T cell-mediated responses.
First author shor biography
Sara Coletta is a 3rd year PhD student in Marina de Bernard’s laboratory. Sara’s project consists in understand whether CD300e can be considered an immune check point and whether it is a part of immune escape strategies occurring in some bacterial infection (i.e. Helicobacter pylori). After BSc in Biotechnology, Sara performed master studies in Industrial Biotechnologies. Before starting the PhD she won an AIRC Fellowship to study the Nuclear DNA repair and mitochondrial DNA maintenance in human SAMHD1 mutant fibroblast in Chiara Rampazzo’s Lab.





